Pharmaceutical logistics is far more sensitive than standard shipping because every shipment can directly impact patient safety, product integrity, and regulatory compliance. Unlike regular parcels, pharmaceutical products often come with strict handling, documentation, and transit requirements. This is especially true for vaccines, specialty drugs, biologics, and temperature-sensitive medicines that move across cities, countries, and multiple logistics partners. In this environment, Pharmaceutical Shipment Tracking becomes essential for maintaining shipment precision, visibility, and accountability throughout transit.
While specialized logistics providers handle temperature-controlled transportation and cold chain infrastructure, shipment monitoring and visibility are equally important. A delay at customs, a missed carrier scan, or a failed delivery handoff can create operational blind spots that impact compliance and business continuity. In pharmaceutical logistics, shipping precision matters just as much as storage conditions.
Market Trends Driving Change in Pharmaceutical Logistics
The pharmaceutical logistics industry is evolving rapidly as global demand for specialty medicines, biologics, vaccines, and personalized treatments continues to rise. These products require stricter transportation workflows, stronger documentation processes, and better shipment coordination across global supply chains.
According to a recent report by Grand View Research, the global pharmaceutical logistics market was valued at USD 99.33 billion in 2024 and is projected to reach USD 208.26 billion by 2033, growing at a CAGR of 8.5%. The report highlights that rising demand for temperature-sensitive drugs, expanding global pharmaceutical trade, and increasing adoption of track-and-trace technologies are major drivers accelerating logistics modernization.
At the same time, growing cross-border pharmaceutical shipments have increased operational complexity across customs clearance, multi-carrier handoffs, and last-mile delivery coordination. This has made shipment visibility a growing operational priority, even when cold storage and temperature management are handled by specialized logistics providers.
As pharmaceutical supply chains become more distributed and compliance expectations continue to tighten, businesses are investing in systems that improve shipment tracking accuracy, milestone monitoring, and faster exception management.
Compliance Challenges in Pharmaceutical Shipment Tracking and Documentation
Pharmaceutical supply chains operate under strict regulations that require complete traceability, accurate documentation, and auditable shipment records. Regulatory frameworks such as Good Distribution Practice (GDP) establish standards to ensure pharmaceutical products are transported under controlled and compliant conditions.
For pharmaceutical companies, compliance is not limited to product handling alone. It also includes maintaining clear records of shipment movement, delivery confirmations, proof of handoffs, and documentation that can support audits or regulatory reviews.
GDP Compliance and Shipment Traceability
GDP guidelines require businesses to maintain control over product movement from origin to destination. This includes having accurate shipment histories, carrier records, and documented handoffs throughout transit.
Without strong tracking systems, businesses often struggle to answer operational questions such as:
- Where is the shipment currently located?
- Was the shipment handed over to the correct carrier?
- Did the delivery occur within the expected timeline?
- Were there unexpected transit delays?
Lack of shipment traceability creates compliance risks and operational uncertainty.
Documentation and Audit Readiness
Pharmaceutical businesses are expected to maintain shipment documentation for internal reviews, customer inquiries, and regulatory audits.
Important shipment records often include:
- Shipping labels and invoices
- Carrier manifests
- Proof of dispatch
- Delivery confirmations
- Shipment milestone logs
- Exception and delay records
Managing these records manually across multiple carriers can quickly become complex, especially when shipment volumes increase.
Missing documentation or incomplete shipment records can lead to delayed audits, compliance gaps, and reduced operational confidence.
Chain of Custody and Proof of Movement
Pharmaceutical shipments frequently pass through several stakeholders, including warehouses, transport partners, customs teams, and last-mile carriers.
Every movement creates a handoff point that must be tracked and documented.
Common challenges include:
- Missing carrier scans
- Delayed milestone updates
- Incomplete handoff records
- Unclear shipment ownership during transit
Without clear proof of movement, businesses may struggle to investigate delays or validate shipment timelines.
Communication Gaps Across Teams
Pharmaceutical logistics often involves coordination between operations teams, customer service teams, compliance managers, third-party logistics providers, and distributors.
When shipment data is fragmented, teams rely on:
- Manual carrier follow-ups
- Email-based status updates
- Spreadsheet tracking
- Delayed escalation processes
This slows decision-making and increases the risk of missed compliance milestones.
Real-time tracking helps businesses maintain accurate shipment histories, improve shipment visibility, and reduce compliance blind spots across logistics workflows.
![]()
How Real-Time Tracking Improves Pharmaceutical Shipment Visibility
Cold chain transport may be managed by specialized logistics providers, but pharmaceutical companies still require end-to-end awareness of shipment movement.
This is where real-time shipment tracking becomes operationally critical.
Instead of waiting for delayed carrier responses or manually checking shipment statuses, businesses can monitor shipments continuously through centralized tracking systems.
Live Shipment Tracking Across Carriers
Pharmaceutical shipments often move through multiple logistics partners across domestic and international routes.
A centralized visibility layer allows teams to track shipments across carriers from one interface, reducing the need to log into separate portals.
This provides:
- Unified shipment monitoring
- Faster access to shipment status
- Improved operational consistency
Multi-carrier visibility becomes especially valuable during large-scale or cross-border pharmaceutical distribution.
Milestone-Based Shipment Updates
Pharmaceutical operations rely heavily on milestone accuracy.
Important milestones include:
- Order dispatched
- In transit
- Customs clearance
- Out for delivery
- Delivered
Missing or delayed milestone updates can disrupt downstream operations and customer communication.
Milestone-based tracking allows teams to monitor shipment progress at each stage and identify delays early.
Predictive ETAs and Delay Alerts
Shipment delays are common in pharmaceutical logistics due to:
- Customs hold-ups
- Carrier congestion
- Documentation checks
- Route disruptions
Waiting until a shipment is overdue creates reactive workflows.
With predictive shipment alerts, businesses can receive early warnings when shipments are likely to miss delivery windows.
This allows operations teams to:
- Notify customers proactively
- Escalate with carriers faster
- Adjust downstream planning
Predictive ETAs improve decision-making by shifting teams from reactive follow-up to proactive intervention.
Exception Notifications for Operational Risks
Operational issues rarely begin with complete shipment failures. Most problems start as small exceptions.
Examples include:
- Missed carrier scan
- Route deviation
- Delayed warehouse processing
- Failed delivery attempt
- Delayed last-mile handoff
Automated exception notifications help teams identify risks early and act before issues escalate.
For pharmaceutical businesses, this level of delivery visibility reduces uncertainty and improves shipment accountability.

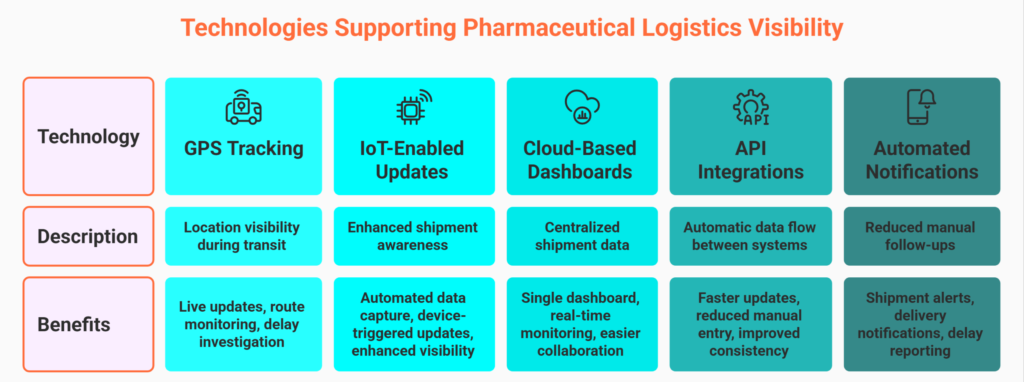
Technologies Supporting Better Pharmaceutical Logistics Visibility
Modern pharmaceutical logistics depends on digital systems that improve shipment coordination, tracking accuracy, and stakeholder communication.
While temperature infrastructure is managed externally, software platforms create the visibility layer required to monitor and manage shipment performance.
GPS Tracking and Location Monitoring
GPS-based shipment tracking provides location visibility during transit.
Benefits include:
- Live shipment location updates
- Improved route monitoring
- Better delay investigation
GPS visibility helps teams quickly identify shipment slowdowns or route anomalies.
IoT-Enabled Shipment Updates
IoT technologies are increasingly used in pharmaceutical logistics to enhance shipment awareness.
These systems can support:
- Automated shipment data capture
- Device-triggered shipment updates
- Enhanced logistics visibility
Even when businesses do not directly manage sensor infrastructure, IoT-driven data can improve operational awareness.
Cloud-Based Visibility Dashboards
Cloud platforms centralize shipment data from carriers, warehouses, and logistics partners.
Benefits include:
- Single dashboard visibility
- Real-time shipment monitoring
- Easier operational collaboration
Centralized dashboards reduce dependency on manual tracking workflows.
API Integrations with Carriers
Pharmaceutical businesses often work with multiple logistics providers.
API-based carrier integrations allow shipment data to flow automatically between systems.
This supports:
- Faster status updates
- Reduced manual data entry
- Improved shipment consistency
Automated integrations create scalable tracking operations.
Automated Notifications and Shipment Analytics
Automated workflows reduce the need for manual shipment follow-ups.
Key capabilities include:
- Shipment alerts
- Delivery notifications
- Delay reporting
- Performance analytics
Using shipment analytics, teams can evaluate carrier performance, identify recurring delays, and improve logistics decision-making over time.
These technologies improve:
- Faster exception management
- Better stakeholder coordination
- Reduced operational delays
Building a More Reliable Pharmaceutical Shipping Strategy with Visibility
Pharmaceutical supply chains require more than transportation execution. They need operational resilience, compliance readiness, and shipment control at scale.
As pharmaceutical businesses expand globally and shipment volumes increase, logistics complexity grows alongside regulatory expectations.
A stronger shipping strategy should focus on:
- Scalable tracking infrastructure
- Accurate shipment records
- Proactive exception handling
- Better customer communication
- Reduced dependency on manual updates
Shipment visibility supports both operational efficiency and compliance preparedness.
When teams have access to centralized shipment data, they can:
- Respond faster to disruptions
- Maintain stronger audit trails
- Improve stakeholder communication
- Reduce tracking blind spots
The future of pharmaceutical logistics will increasingly depend on integrated ecosystems where transportation partners manage cold chain movement, while digital platforms provide logistics visibility solutions, tracking consistency, and operational control.
In an industry where timing, documentation, and accountability directly impact product quality, shipment visibility is no longer optional. It is a critical part of building a reliable pharmaceutical shipping operation.
